 
Each proton carries an electric charge of +1, so the atomic number also specifies the electric charge of the nucleus. It is, rather, the number of protons in the nucleus, which we call the atomic number and denote by the symbol Z. What single parameter uniquely characterizes the atom of a given element? It is not the atom's relative mass, as we will see in the section on isotopes below. The radius of an atom must be defined arbitrarily, such as the boundary in which the electron can be found with 95% probability. The latter has no definite outer boundary, so neither does the atom. It is common (but somewhat misleading) to describe the volume of space in which the electrons of an atom have a significant probability of being found as the electron cloud. 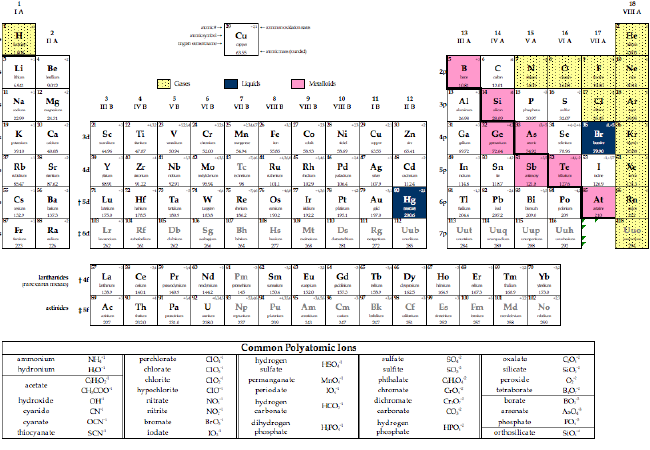
The extremely small mass of the electron (1/1840 the mass of the hydrogen nucleus) causes it to behave as a quantum particle, which means that its location at any moment cannot be specified the best we can do is describe its behavior in terms of the probability of its manifesting itself at any point in space. 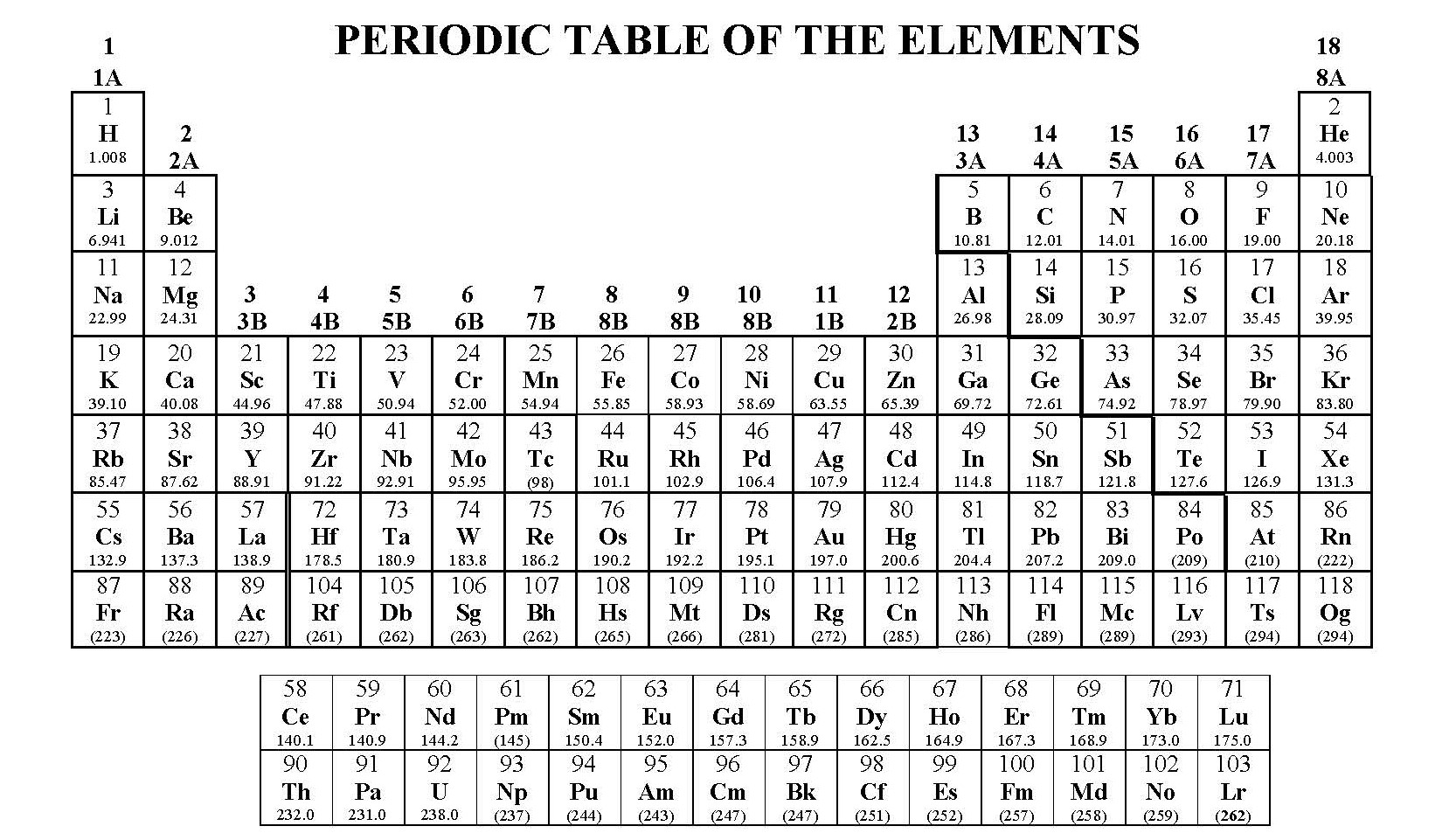
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
